VASCULAR DISEASE: IT STARTS IN THE WALL
Early Detection of Vascular Disease through Arterial Waveform Analysis
by Dr. Jay N. Cohn, M.D. Professor of Medicine University of Minnesota Medical School Cardiovascular Division Minneapolis, Minnesota
Dr. Cohn founded the Heart Failure Society of America and was its first president and also founded the Journal of Cardiac Failure.
- Director, Rasmussen Center for Cardiovascular Disease Prevention
- President, International Society of Cardiovascular Pharmacotherapy (2008-2010)
- Scientific Advisory Committee (1996-1998)
- Victor Chang Cardiac Research Institute, Sydney, Australia (1997)
- President, International Society of Hypertension (1996)
SUMMARY
Significant advances have occurred in the prevention and treatment of vascular disease. Yet vascular disease remains as the leading cause of death in the United States. Awareness of risk factors has encouraged healthier eating habits and more active lifestyles. Risk factors can include:
- smoking
- inactivity
- obesity
- diabetes
- high blood pressure
- high cholesterol
- genetics
Prevention efforts target the entire population or those with specific risk factors. The whole- population approach imposes lifestyle alterations on a large population. Yet, only some of whom are at risk would realize such changes. The so-called high-risk population may or may not ever develop vascular disease.
Hypertension is present in less than 40 percent of patients sustaining a heart attack. High cholesterol levels are present in less than 40 percent of patients as well. This approach is useful. But, it does not identify the actual presence of vascular disease in its early stages. Early identification with current techniques is rare due to cost and other factors.The University of Minnesota developed an advanced technology, called Arterial Waveform Analysis. This holds the promise of detecting vascular disease at the earliest stages. Subtle changes in the arterial wall can provide early markers for vascular disease.Arterial waveform analysis may also help physicians improve management of the pharmacological therapy. This can aid in efforts to better use healthcare resources. Also, early intervention and life style changes are especially useful.
UNDERSTANDING THE PROBLEM – WHO REALLY IS AT RISK?
Vascular disease accounts for one-half of all deaths in the United States each year. It is the primary cause of heart attacks and strokes. Vascular disease can manifest itself in many ways: hypertension, coronary artery disease, peripheral artery disease, atherosclerosis, aneurysm, stroke, kidney failure and retinopathy. According to the AMA, 58 million Americans have some form of cardiovascular disease. Vascular disease can manifest itself in many ways:
- hypertension
- cornonary heart disease
- periphreal artery disease
- atherosclerosis
- aneurysm
- kidney failure
- retinopathy
HYPERTENSION
Hypertension is the leading cause of cardiovascular disease. Coronary artery disease affects 13.9 million Americans and is the nation’s number one killer. Stroke is the number three killer.According to a 1997 release by the National Heart, Lung and Blood Institute, 50 million adults in the US are diagnosed with hypertension. Hypertension is defined as blood pressure greater than 140/90. Almost 75% of those are not properly treated for the condition. Those who are not treated face significant increased risk for heart disease.According to the Johns Hopkins White Papers on Hypertension (1998), 30 million Americans have “high- normal hypertension.” This is also referred to “borderline hypertension.” It’s defined as a blood pressure reading at or slightly above 130/85.These individuals are twice as likely to develop hypertension. They have a greater risk of CDV events than people with lower blood pressure. High-normal blood pressure is common in the United States. The majority of CV events occur because high blood pressure happens in people who have the condition but aren’t treated for it.Hypertension is of particular concern to older adults, as levels increase with age. It accounts for 15-20% of all deaths over fifty years of age in the United States. Women also appear to be at increased risk with 33% of deaths caused by heart disease. 17% are relatable to stroke. Half of the women affected by stroke exhibited no symptoms of cardiovascular disease.HYPERTENSION – THE SILENT KILLER
Since hypertension can go undetected, it’s referred to as the “silent killer.” It usually produces no symptoms until after damage occurs to the heart, kidneys, or brain. This problem is more serious as the population grows older. Inviduals with high blood pressure have an increased death rate. This can be from stroke, heart attacks, or kidney disease.
The standard test for screening for vascular disease is use of an upper-arm blood pressure cuff device. While the cuff yields some information, the information it provides is very limited. It doesn’t note the presence of blood vessel disease. The blood pressure reading can be variable. It can be influenced by many factors, including physical activity, stress, etc.When the aortic heart valve closes after ejecting its stroke volume, the decay of blood pressure before the next heartbeat describes a waveform which is dependent on the stiffness of the arterial system into which the blood is delivered.The data from the blood pressure waveform can tell a lot about the likelihood of atherosclerosis. With this information, treatment may start for suspected CVD for patients with one or more risk factors. It is also possible that many of these patients do not have the disease. Some patients who develop atherosclerosis don’t exhibit high risk symptoms. Because of this, they may not receive treatment.Augmenting current prevention efforts with positive identification of blood vessel disease has the potential to yield significant advances. Screening and diagnosing patients for CVD, the incidence and severity of heart attacks and strokes can reduce.
Get Your Max Pulse Today!
Purchase with 5 Year Warranty – As Low As $240 a Month!
(Includes Max Pulse® with Accessories, and Laptop with Software.
There are no other ongoing fees or costs)
Call: (210) 610-2521 or fill out the form below for More Info
VASCULAR DISEASE: IT STARTS IN THE WALL
Hypertension is a deadly disease that damages both large and small arteries. This leads to pathological changes in the tissues or organs supplied by these damaged arteries. It also accelerates the development of atherosclerosis in large blood vessels. The arteries supplying blood to the brain, heart, kidneys, and legs are also affected.Plaques can cause minimal strokes (transient ischemic attacks) due to diminished blood flow (ischemia) to parts of the brain. Angina occurs from partly obstructed coronary arteries. Pain in the leg muscles when walking can be a result of poor blood supply to the legs (peripheral arterial disease). Blood clots, which occur at the sites of atherosclerotic narrowing. This can block a vessel and cause a stroke or heart attack.The walls of all arteries (large and small) throughout the body are distensible. They expand and contract as blood pressure waves from the heart pass through the lumen. This is the passageway for blood within the arterial walls.The distensibility enables the arterial system to act as an elastic reservoir that stores part of the energy of each cardiac contraction. This maintains blood pressure and flow during diastole to perfuse all tissues.Changes in function and structure of the arterial wall precede the development of obstructive CAD. They are an early sign of the hypersensitive disease process. Vascular changes in elasticity can be induced by changes in:
- the smooth muscle
- the mass of the smooth muscle collagen
- elastin components of the wall
- infiltration of the wall with cellular or interstitial elements
- the change in tissue fluid in the wall
THE WALL
The arterial wall is composed of three concentric zones termed the tunica intima, tunica media, and tunica adventitia. The tunica intima consists of the vascular endothelium. This is a thin layer of collagen and elastin fibers that anchor it to the internal elastin lamina. Recent research has explored the role the endothelium plays in the development of blood vessel disease. If the endothelium is thought of as an internal organ, it would be one of the largest organs of the body. Spread over a flat area, the endothelium cells lining the arteries of a single adult would cover the area of three football fields. Nitric oxide gas is released by the endothelium cells, and relaxation of the arterial walls is believed to be an important result.
The smallest arteries and arterioles are the sites of greatest hemodynamic resistance. They act in conjunction with the precapillary sphincters. Also, they form a variable resistance that controls the rate of blood flow perfusing all body tissue. An arterial system composed of elastic conduits and high-resistance terminals constitutes a hydraulic filter that converts the intermittent output from the heart into steady capillary flow.THE ARTERIAL BLOOD PRESSURE WAVEFORM
The arterial blood pressure waveform is from the complex interaction of the left ventricle stroke volume, the physical properties of the arterial tree and the characteristics of the fluid (blood) in the system. Changes in arterial vascular compliance introduce changes in the arterial system. These reflect in the arterial blood pressure waveform. Changes are seen when examining the difference in the arterial pressure waveform of a patient before and after the administration of a pharmacological agent such as nitropurusside or nitroglycerin.During systole, only the proximal part of the aorta becomes distended at first. This is because the inertia of blood hinders the passage of the total stroke volume to the periphery. The radial stretch of the ascending aorta brought about by left ventricle (LV) ejection initiates a blood pressure wave. This propagates down the aorta and its branches. This pressure wave moves at a finite velocity that is faster than the actual forward movement of the blood itself. The pressure pulse is simply a moving wave of pressure that involves very little forward movements of blood.The distortion in the arterial waveform includes a delay in the time of the onset of the initial pressure rise. This dampens the high- frequency components of the pulse, and a narrowing and elevation of the systolic portions of the pressure wave.In the initial portion of the diastolic pressure waveform, a feature referred to as the “dicrotic notch”becomes more prominent as the pulse passes peripherally. These morphologic changes tend to diminish with age as the arteries lose their elasticity. The damping of the high-frequency components of the arterial pulse are due to the viscoelastic properties of the arterial walls. The mechanisms involved in the peaking of the pressure wave are not fully understood.ARTERIAL WAVEFORM ANALYSIS
Functional and structural changes in the arterial wall precede the development of obstructive CAD. They may be early markers for the hypertensive disease process. Vascular (or arterial) compliance has long been recognized as declining as part of the normal aging process. Also, as a declining at an accelerated rate in people with heart disease. The aorta and large arteries can be visualized by various x-ray and other techniques. Yet, there has been no means to test the flexibility or elasticity of the small arteries throughout the body. These can become altered in hypertension and with other vascular diseases.Cardiovascular specialists spend considerable effort on evaluating heart function. including electrocardiograms (EKGs), echocardiograms and stress tests. But, they have been unable to assess the functional and structural abnormality of the arteries prior to the late phase of arterial obstruction (as determined by angiography).THE WINDKESSEL MODEL
The arterial waveform analysis method utilizes a modified Windkessel model. This describes the pressure changes during the diastolic phase of the cardiac cycle in the circulatory system. This analysis method provides an assessment of the elasticity or flexibility of the large arteries. These expand to store blood ejected by the heart, and of the small and very small arteries (arterioles) which produce oscillations or reflections in response to the blood pressure waveform generated during each heartbeat.Assessing the elasticity of the arterial system, clinical investigators have been able to identify a reduction in arterial elasticity in patients without evidence of traditional risk factors. This suggests the early presence of vascular disease. Research has demonstrated that individuals with heart failure, coronary artery disease, hypertension and diabetes exhibit a loss of arterial elasticity. These abnormal blood vessel changes often appear to precede overt signs of CVD and the occurrence of a heart attack or stroke by many years. Clinical investigators have also demonstrated an age- related loss of elasticity of both the large and small arteries. This suggests that premature stiffening of an individual’s arteries is a marker for the early onset of CVD.Dozens of clinical research studies and pharmaceutical trials have been completed by utilizing this technology. More than 2,500 subjects have been reviewed. This has resulted in more than 30 scientific articles published in peer-reviewed medical journals.Dr. Jay N. Cohn, M.D
(Contact Us to Obtain Copies of the Most Important Scientific Studies)
Get Your Max Pulse Today!
Purchase with 5 Year Warranty – As Low As $240 a Month!
(Includes Max Pulse® with Accessories, and Laptop with Software.
There are no other ongoing fees or costs)
Call: (210) 610-2521 or fill out the form below for More Info
Contact Us:
We would love to hear from you! Please fill out this form and we will get in touch with you shortly.
 Vascular disease
Vascular disease  Since hypertension can go undetected, it’s referred to as the “silent killer.” It usually produces no symptoms until after damage occurs to the heart, kidneys, or brain. This problem is more serious as the population grows older. Inviduals with high blood pressure have an increased death rate. This can be from stroke, heart attacks, or kidney disease.
Since hypertension can go undetected, it’s referred to as the “silent killer.” It usually produces no symptoms until after damage occurs to the heart, kidneys, or brain. This problem is more serious as the population grows older. Inviduals with high blood pressure have an increased death rate. This can be from stroke, heart attacks, or kidney disease. 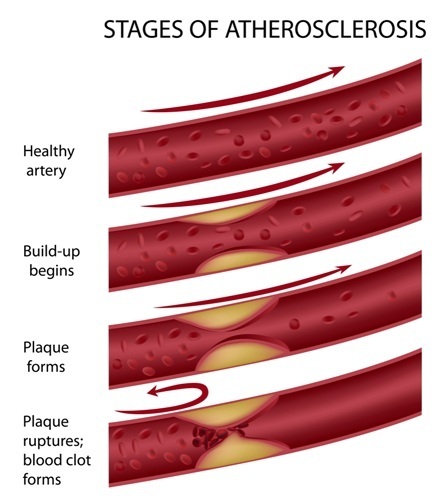 The arterial wall
The arterial wall